|
Hepatoma-derived growth factor (HDGF)-related proteins (HRPs)
comprise a new family of proteins that are mitogenic for various cell lines. Members of this family include
human HDGF (hHDGF), HRP-1, HRP-2, HRP-3, HRP-4, and lens epithelium-derived growth factor, LEDGF/p75/p52.
These proteins share a conserved N-terminal PWWP module (amino acids 1-100) with high sequence identity,
but the C-terminal regions show considerable variation in length and charge. The PWWP module contains a PWWP
motif that is a structural domain frequently found in eukaryotes and plays important roles in cell growth
and differentiation, and in protein–protein interactions.
Human HDGF (hHDGF), the first HRP discovered, was originally isolated from conditioned medium of human
hepatoma-derived Huh7 cells as a heparin-binding growth factor that can stimulate DNA synthesis in Swiss 3T3
cells, and was subsequently found to be widely expressed in various organs, including brain, testis, lung, and
spleen, and distributed in various subcellular compartments, including the nucleus and cytoplasm. It promotes
proliferation of fibroblasts, renal epithelial cells, smooth muscle cells, and fetal hepatocytes, and was
found to play a role in the development of vascular tissue, kidney and liver, and may be involved in lung
remodeling after injury by promoting the growth of lung epithelial cells. Among the growth factors responding
to vascular wall injury, hHDGF is unique in that it is only expressed after injury. This property makes it an
attractive target for therapeutic intervention. In addition, hHDGF was found to be over-expressed in human
gastric cancers and hepatocellular carcinoma, and thus is likely to play an important role in mitogenesis
and tumorigenesis. Recently, the role of hHDGF in the nervous system has been investigated and a role as a
neurotrophic factor has been proposed. Therefore, hHDGF appears to play a role in a wide range of important
biological processes. We have investigated the structure and function of HDGF. Our achievements include:
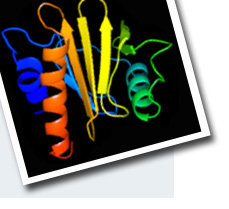
We have demonstrated that hHDGF adopts a novel modular structure for function. It consists of a structured
N-terminal PWWP module (residues 1–100) and a disordered C-terminal domain (residues 101–240). The N-terminal
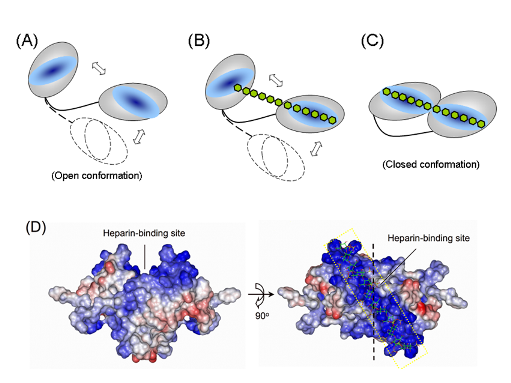
PWWP module is the heparin-binding domain and its structure consists of a five-stranded anti-parallel b-barrel
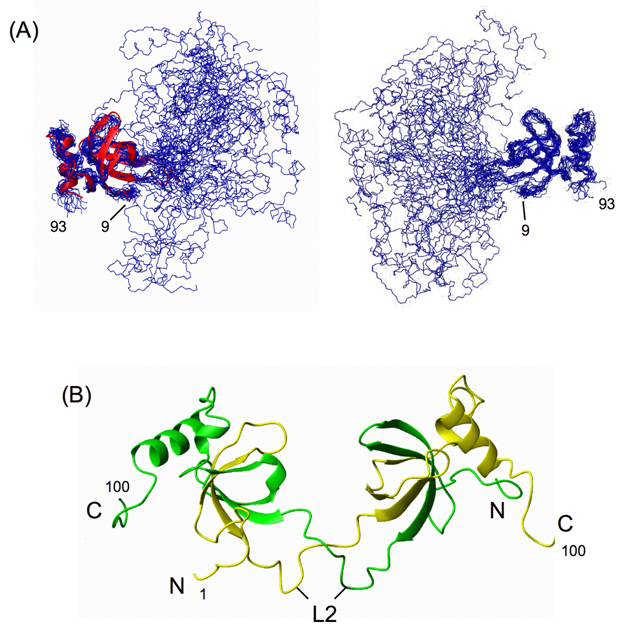
followed by two a-helices. This domain was also shown to bind to DNA.We demonstrate that both hHDGF and its PWWP module can form dimers and that the dimers bind to heparin with affinity two orders of magnitude higher than
that of the corresponding monomers. The PWWP-monomer-dimer equilibrium behavior and NMR structural data suggest that PWWP-dimer formation involves a domain-swapping mechanism.
|
Publications
Sue, S.C., Chang, J.Y. and Huang, T.-h.*
Sequence specific 1H, 13C and 15N resonance assignments of the hath domain of hepatoma derived growth factor. J. Biomol. NMR 29, 95-96.(2004)
Sue, S.C., Chang, J.Y., Lee, S.C., Wu, W. G. and Huang, T.-h.*
Solution structure and heparin binding site of Hepatoma-derived Growth Factors J. Mol. Biol. 343, 1365-1377. (2004)
3. Sue, S.C., Lee, W.T., Tien, S.C., Yu, J.G., Lee, S.C., Wu, W.J., Wu*, W.G. and Huang, T.-h.*
PWWP module of human hepatoma-derived growth factor forms a domain-swapped dimer with much higher affinity for heparin. J. Mol. Biol. 367, 456–472 (2007).
|